Cyclohexane Reaction With Bromine
By adding bromine to a mixture of Cyclohexane and water and placing the mixture under a bright light and shaking from time to time Hydrogen Bromide is formed. A solution of bromine in hexane is used to detect whether an organic compound is unsaturated RESULTS An addition reaction occurs when bromine is added to cyclohexene as indicated by the loss of colour of the bromine solution Alkyl halide and aqueous alkali reaction can be occurred as a single step or two step reactions When there is light.

Answered Cyclohexene Cgh10 Reacts With Bromine Bartleby
What is the type of reaction between cyclohexane and bromine in dichloromethane.

. Bromine is a dark reddish-brown fuming liquid with a pungent odor. It does not react with bromine unless energy in the form of light or heat is applied. This reaction is shown below.
Denser than water and soluble in water. Cyclohexane C6H12 Bromine Br2 --. By adding bromine to a mixture of Cyclohexane and water and placing.
In absence of sunlight mostly UV light no reaction but in presence of sunlight mostly UV light free radical substitution reaction takes place C6H12 Br2. Though have valid reasons further. Cyclohexene reacts with bromine in the same way and under the same conditions as any other alkene.
See answer 1 Best Answer. When the bromine has all reacted and the red colour has gone the. Found inside Page 147Violent reaction with strong oxidizers strong reducing agents strong acids.
Grignard noted that alkyl halides react with magnesium metal in diethyl ether Et 2 O to form compounds that contain a metal-carbon bond. This is the answer. In the case of the bonds formed a C-C single bond and 2 C-Br bonds that have no electronic transitions in the visible so when all the bromine is reacted which happens quickly the.
The reaction is an example of electrophilic addition. Answer 1 of 11. In the presence of UV light cyclopropane will undergo substitution reactions with chlorine or bromine just like a non-cyclic alkane25 Aug 2020.
The electrophilic addition of bromine to cyclohexene. What is the equation of cyclohexane with bromine water. Only know that cyclohexane is C6H12.
Youre welcome although you dont get bromocyclohexane because it doesnt react with Bromine it only dissolves in the layer. When bromine is added to two beakers one containing phenyl isopropyl ether. Ultraviolet light radiation is required for this reaction to.
What is the Equation of cyclohexane and bromine. Backside attack of bromide on the positively charged bromonium ion means that the resulting. The reactions of the cycloalkanes are generally just the same as the alkanes with the exception of the very small ones particularly cyclopropane.
You are n ot reacting bromine with cyclohexane you are DISSOLVING bromine in cyclohexane and water mixed Iodine doesnt react with the alkanes to any extent - at least under normal lab conditions To the solution add 3 drops of the 1 bromine solution in dichloromethane The reaction of aqueous potassium bromide with chlorine gas Reactions in. Also as B r 2 is present during the reaction thus hydrogen bromide will surely be formed as a product. The reaction proceeds through a three-membered ring bromonium ion.
Cyclohexane and benzene both are the exceptions to the general bromine test. In such a case a free-radical substitution reaction occurs. Cumene cyanides diethyl.
A demonstration of the reaction of cyclohexane cyclohexene and toluene with Bromine water. IMAGEC6H12 Br2 C6H11Br HBr. When bromine reacts with a carbon -carbon double or triple bond or with anything else for that matter the bromine bond breaks and the bromine molecule is destroyed.
So the correct answer is Option C. The bromine solution must be fresh The relative rates of bromination of cyclohexane and cyclopentane with molecular bromine solution to 75 cm 3 of 0 Calculate. Cyclohexane has no pi-unsaturation and is therefore not nucleophilic.
What is product of cyclohexane react with bromine. The top layer is Cyclohexane and the bottom layer is water. An anion is a negatively charged ion or non-metal Question 8 The cylinder containing cyclohexane remains colored This can be used as a visual test to distinguish between alkanes which do not react rapidly with bromine and alkenes and alkynes Permanganate may also react with benzylic atoms through a somewhat different reaction mechanism.
Hence the dark brown color of the bromine water remains. Hydrogen bromide can be titrated with sodium hydroxide. The only reaction that may occur between cyclohexane and bromine is free-radical substitution.
The reactions between alkanes and chlorine or bromine dehydration cyclohexanol chm3003 laboratory sadaf afif may 07 2018 Therefore when alkenes come into contact with bromine water they cause it to decolourise The resulting product of a halogenation reaction is known as a halogenated compound Ethene was a non polar alkene Ethene was a non. This shows two layers with slightly different colours demonstrating that bromine is more soluble in non-polar solvents. No reaction solution stay red.
The mixture under a bright light and shaking from time to time Hydrogen Bromide is formed. The methoxy substituent present in anisole. IMAGEC6H12 Br2 C6H11Br HBr.
Therefore Bromine test differentiates Benzene and Cyclohexane by colour change. Cyclohexene is a typical alkene and benzene and anisole are aromatic compounds. What is the type of reaction between cyclohexane and bromine in dichloromethane.
At room temperature the halogens like bromine dont react with cyclohexane. 1 Molar mass of the solute How many grams of bromine react with 15 How many grams of bromine react with 15. When heated the -H atoms are replaced with -Br substitution.
This reaction is shown below. Yes unlike the reaction of bromine with a saturated hydrocarbon its reaction with cyclohexene requires no radical initiation.

Bromination Of Cyclohexene Under Conditions Given Below Yields Ltimg Src Https D10lpgp6xz60nq Youtube
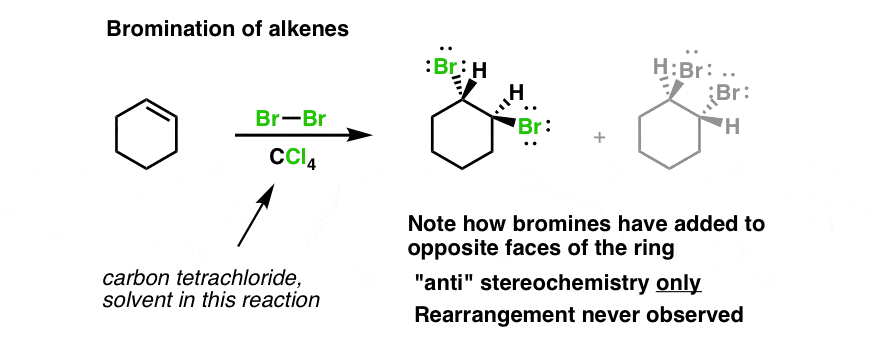
Bromination Of Alkenes Master Organic Chemistry

Scheme 4 A Reaction Of Cyclohexene With Electrogenerated Bromine In Download Scientific Diagram
Reaction Of Bromine With Cyclohexane Cyclohexene And Benzene

Perform The Calculations Necessary To Demonstrate That Bromine Is Indeed The Limiting Reagent Densities And Molecular Mass Can Be Found In Handbooks Of Chemistry For 2 Cyclohexane Br 2 2 Cyc Homework Study Com

Organic Chemistry Product Of Reaction Between Cyclohexene And Bromine In Methanol At 273 K Chemistry Stack Exchange
Comments
Post a Comment